Our Research
Summary
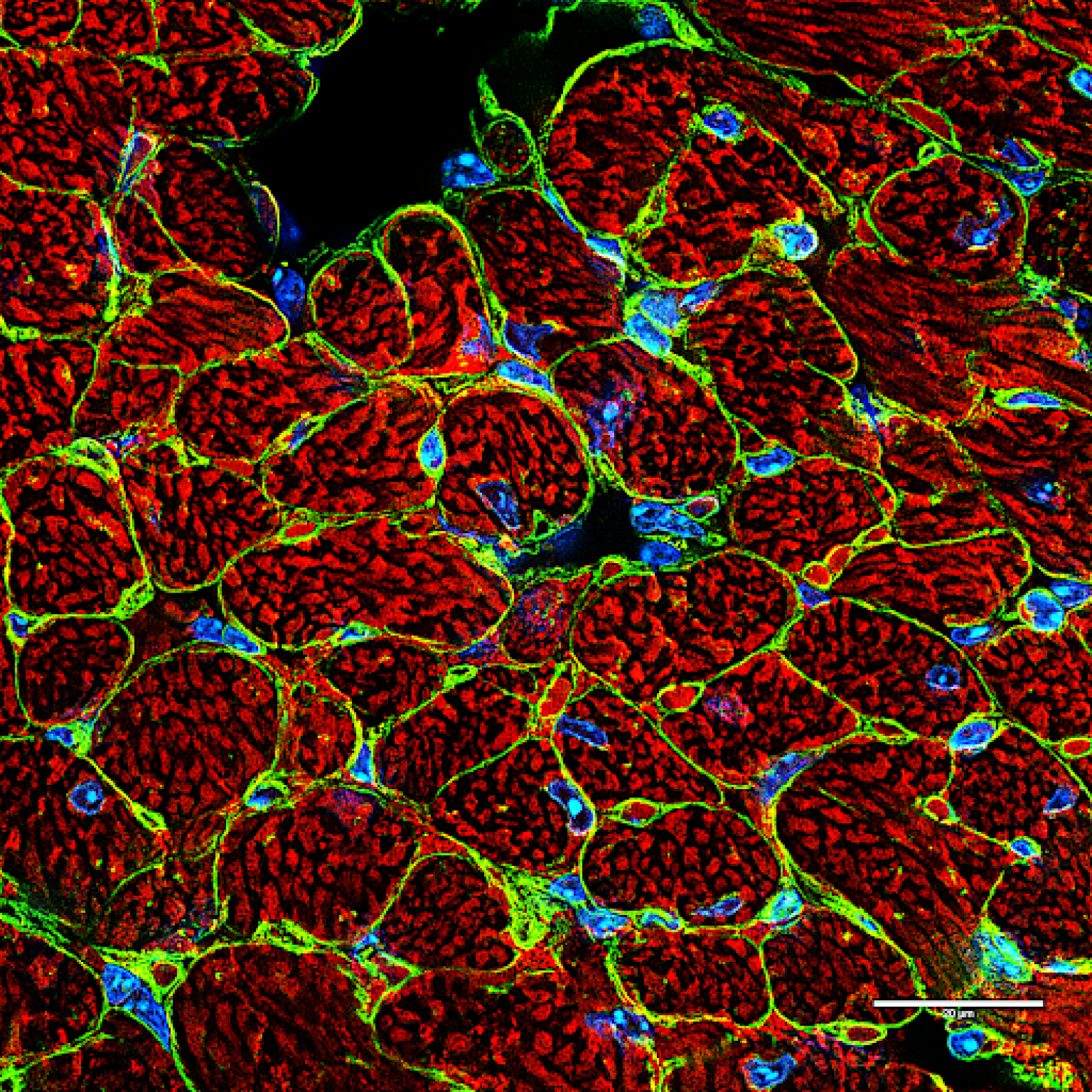
The occurrence of cardiovascular disease and heart failure development increases with
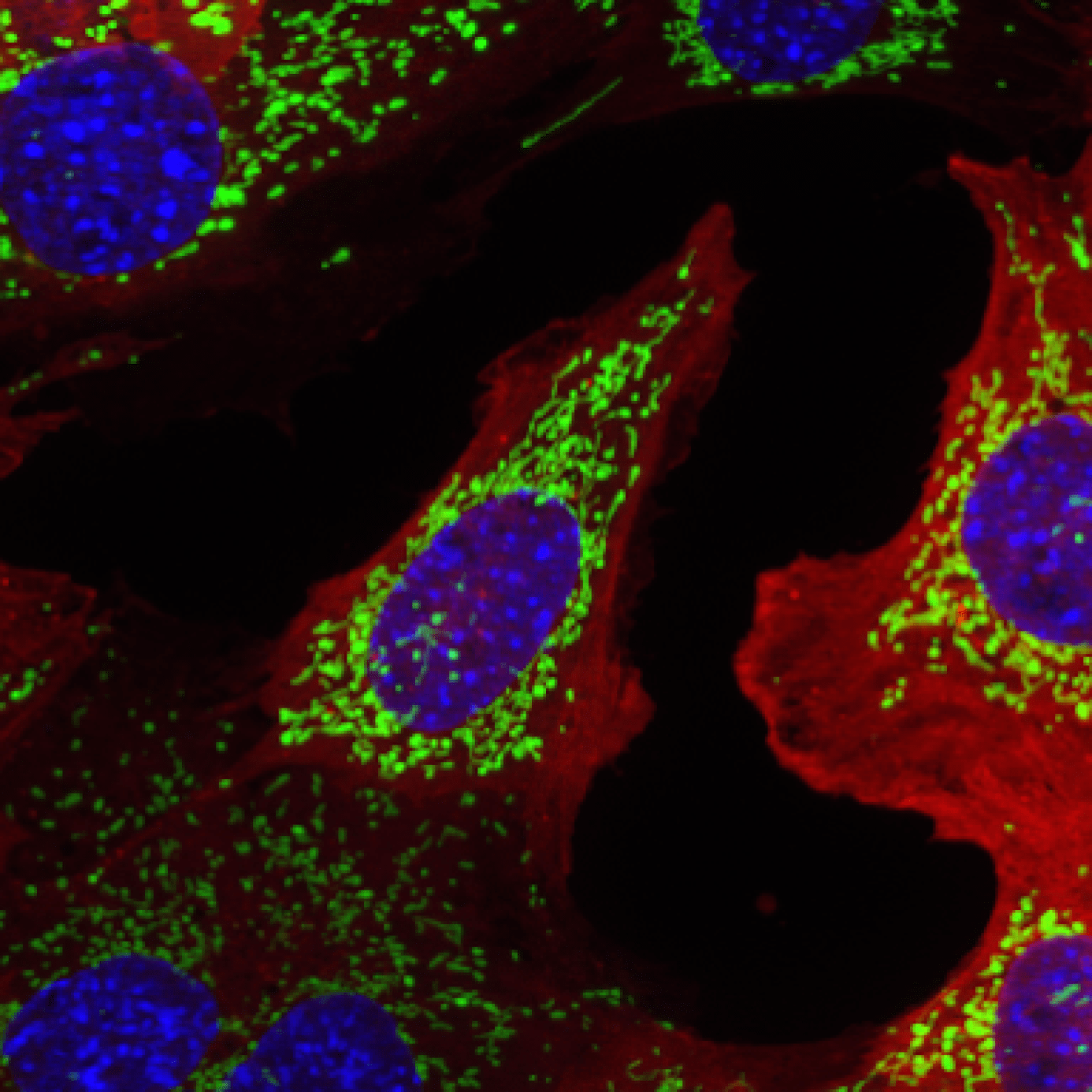
advancing age and intrinsic alterations in aging cardiac myocytes are a major contributor to the underlying pathogenesis. In particular, a decline in mitochondrial function is considered to play a key role in the increased susceptibility to disease. In the heart, the primary function of mitochondria is to meet the high energy demand of the beating myocytes by providing ATP through oxidative phosphorylation. However, mitochondria can quickly change into death-promoting organelles that generate excessive levels of reactive oxygen species and release pro-death proteins. Mitochondrial dysfunction and activation of cell death pathways are common occurrences in cardiovascular disease and contribute to the development of heart failure. Our research is focused on dissecting signaling pathways involved in ensuring mitochondrial health in cells. We are using genetic, cell and molecular biology approaches, mouse models, and cutting-edge 2D/3D imaging techniques to study the signaling pathways involved in regulating mitochondrial structure, function, and turnover in cells and heart.
Autophagy and mitochondria:
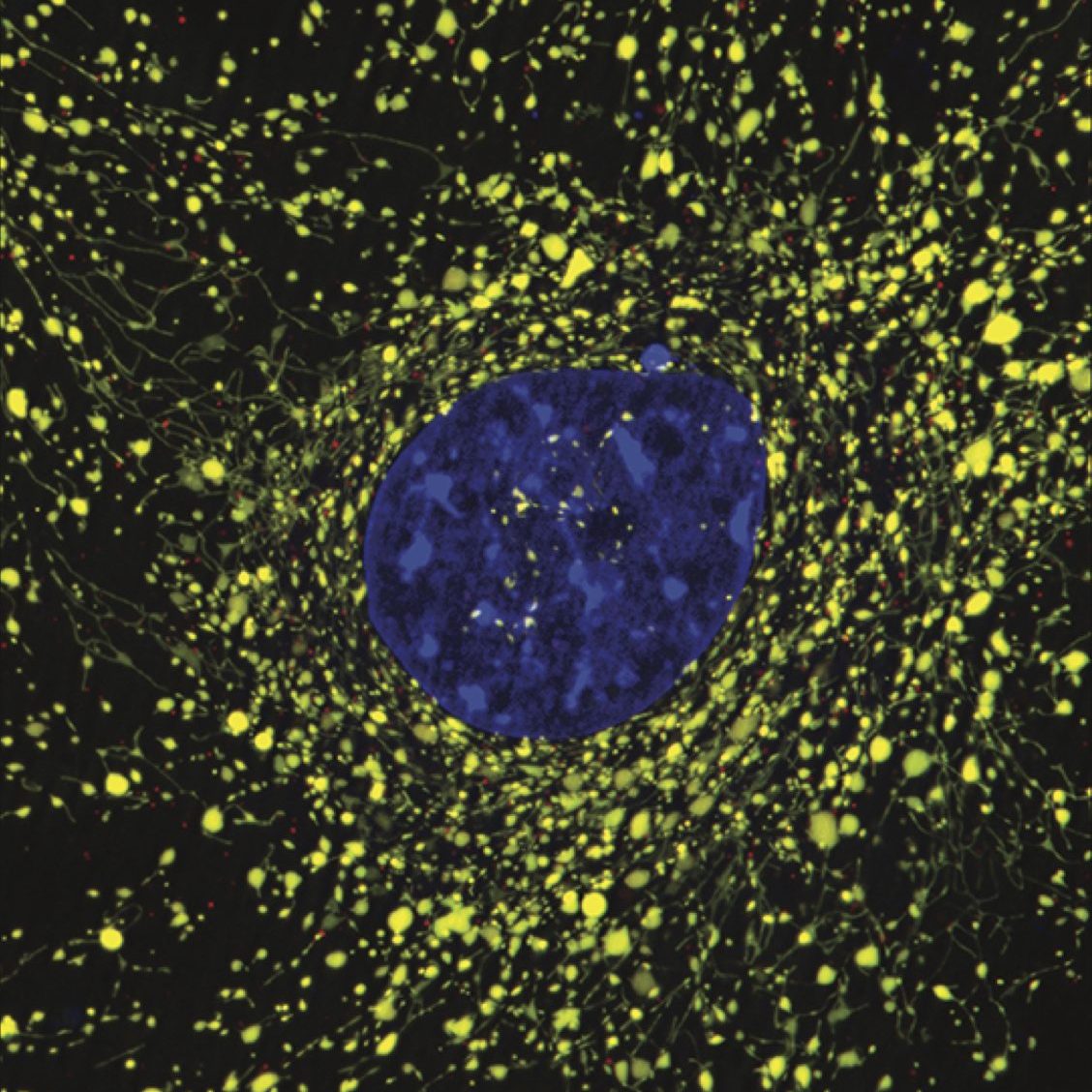
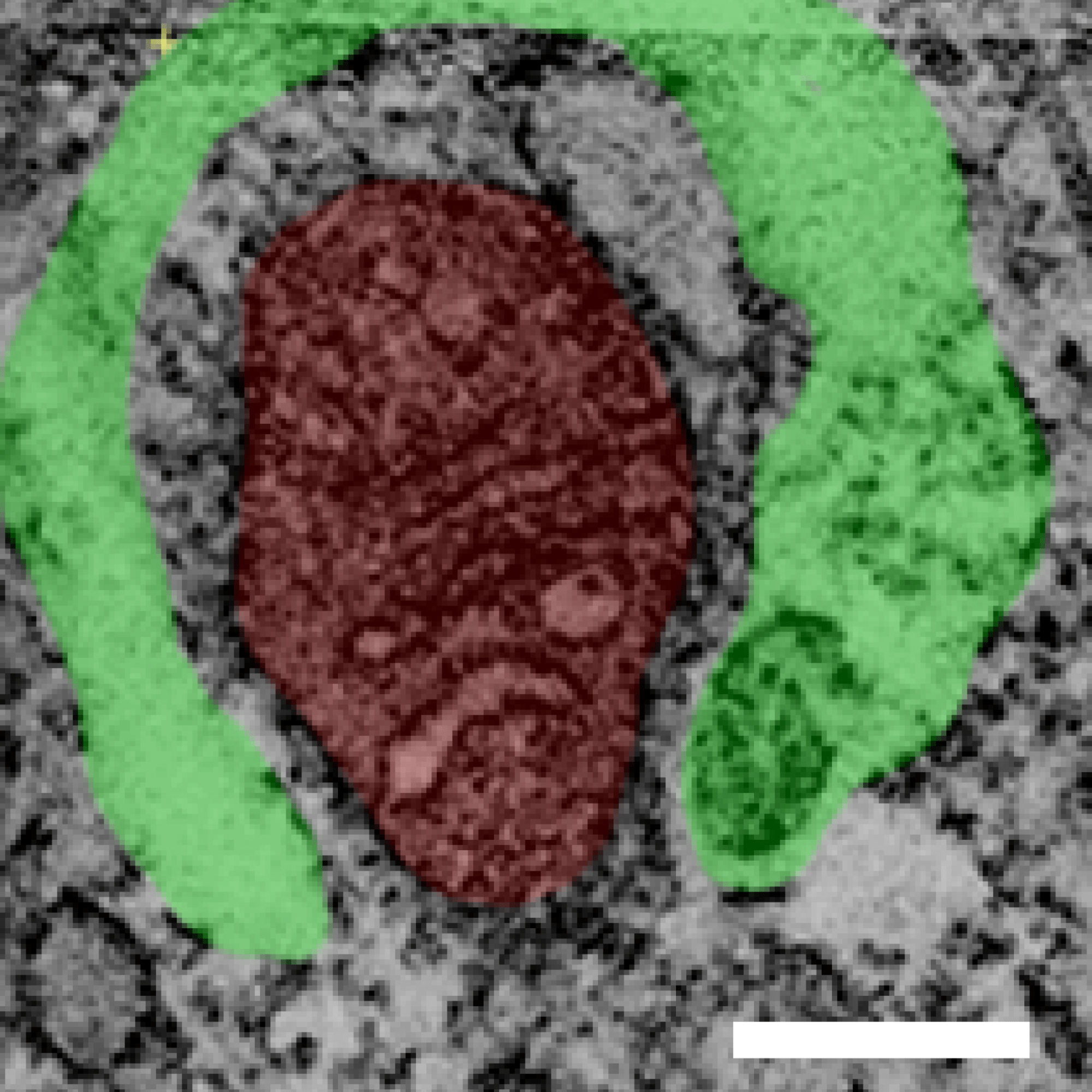
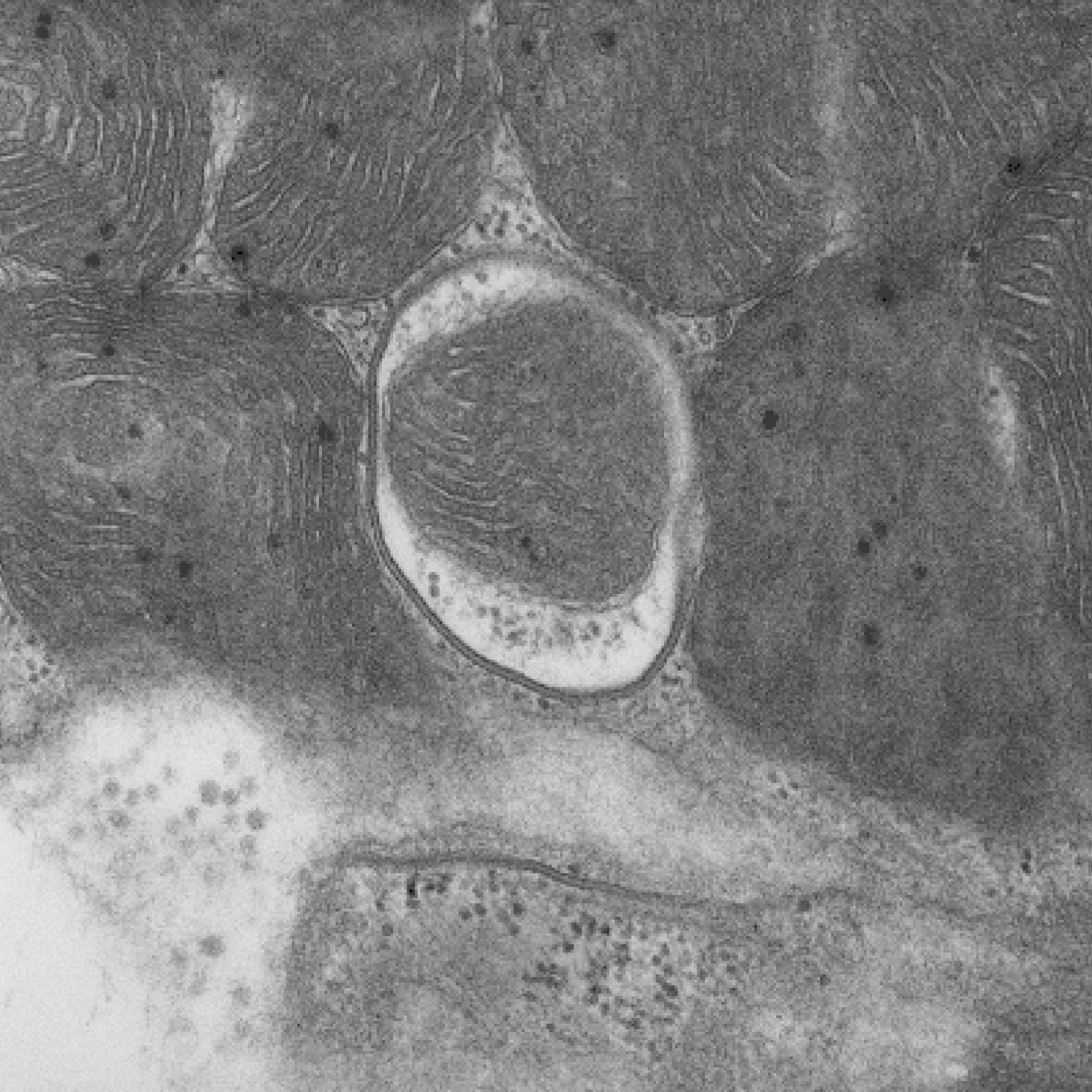
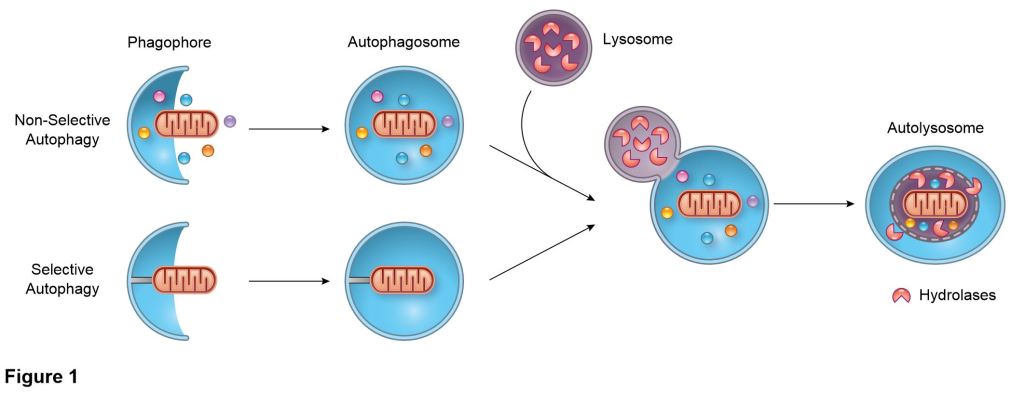
Due to the importance of maintaining a healthy population of mitochondria, cells have developed multiple defense mechanisms against aberrant mitochondria that can cause harm. The ability of a cell to repair itself and prevent unnecessary death is particularly important in a post-mitotic cell such as a myocyte that cannot be easily replaced. During mitochondrial autophagy (also known as mitophagy), dysfunctional mitochondria are rapidly sequestered by autophagosomes and subsequently delivered to lysosomes for degradation (Figure). We are examining the signaling pathways and proteins involved in the identification and labeling of
damaged mitochondria, and the formation of vesicular structures (autophagosomes) required to
sequester the mitochondrion. We are interested in how mitophagy is regulated by the PINK1/Parkin pathway and mitophagy receptors.
Alternative mitochondrial quality control pathways:
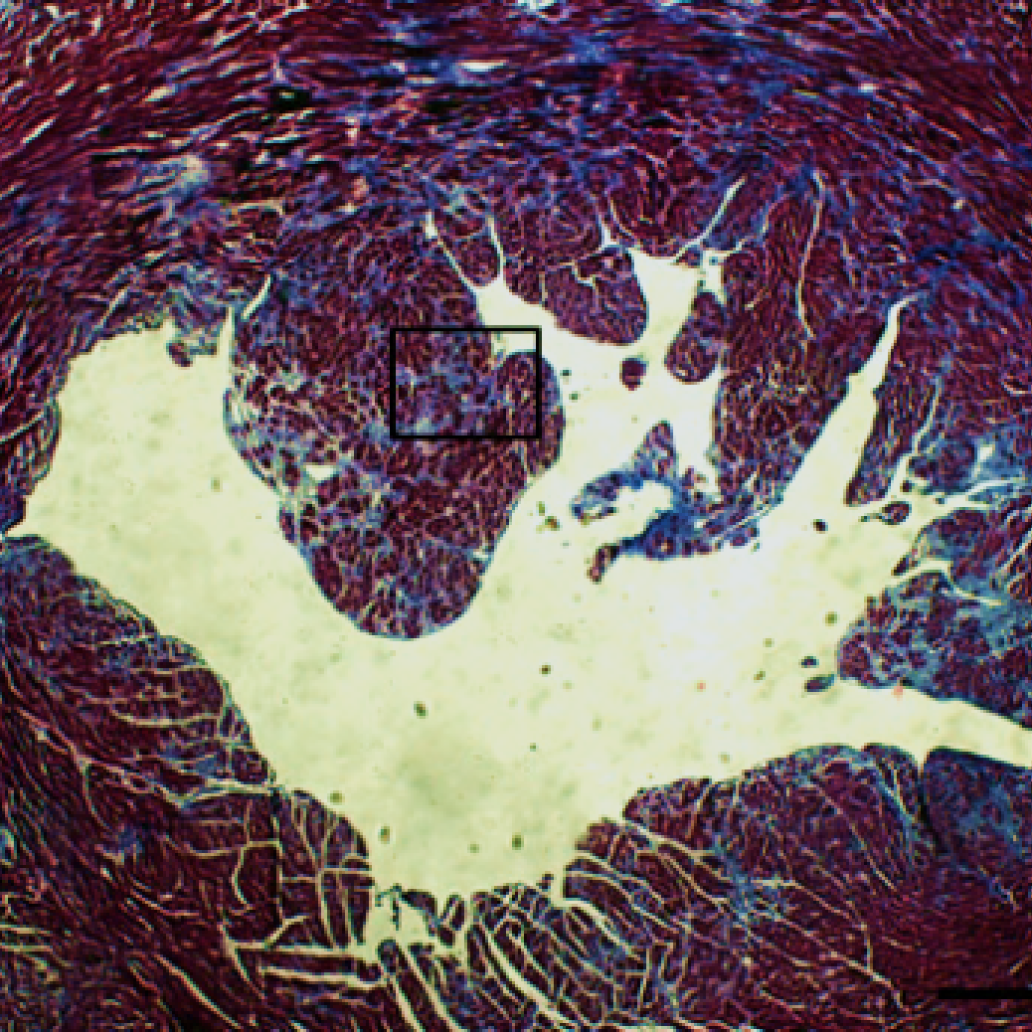
Recent studies suggest that alternative pathways to traditional autophagy exist that can clear unwanted mitochondria. We have uncovered that dysfunctional mitochondria can also be eliminated by Rab5-positive early endosomes and that this pathway is independent of autophagy. The early endosomes mature into late endosomes which then deliver the cargo to lysosomes for degradation. We are currently focused on dissecting the role of Beclin1 and the various Rab GTPases in regulating this process.
Gallery